Certificates Of Analysis For
USP, NF And EP Testing
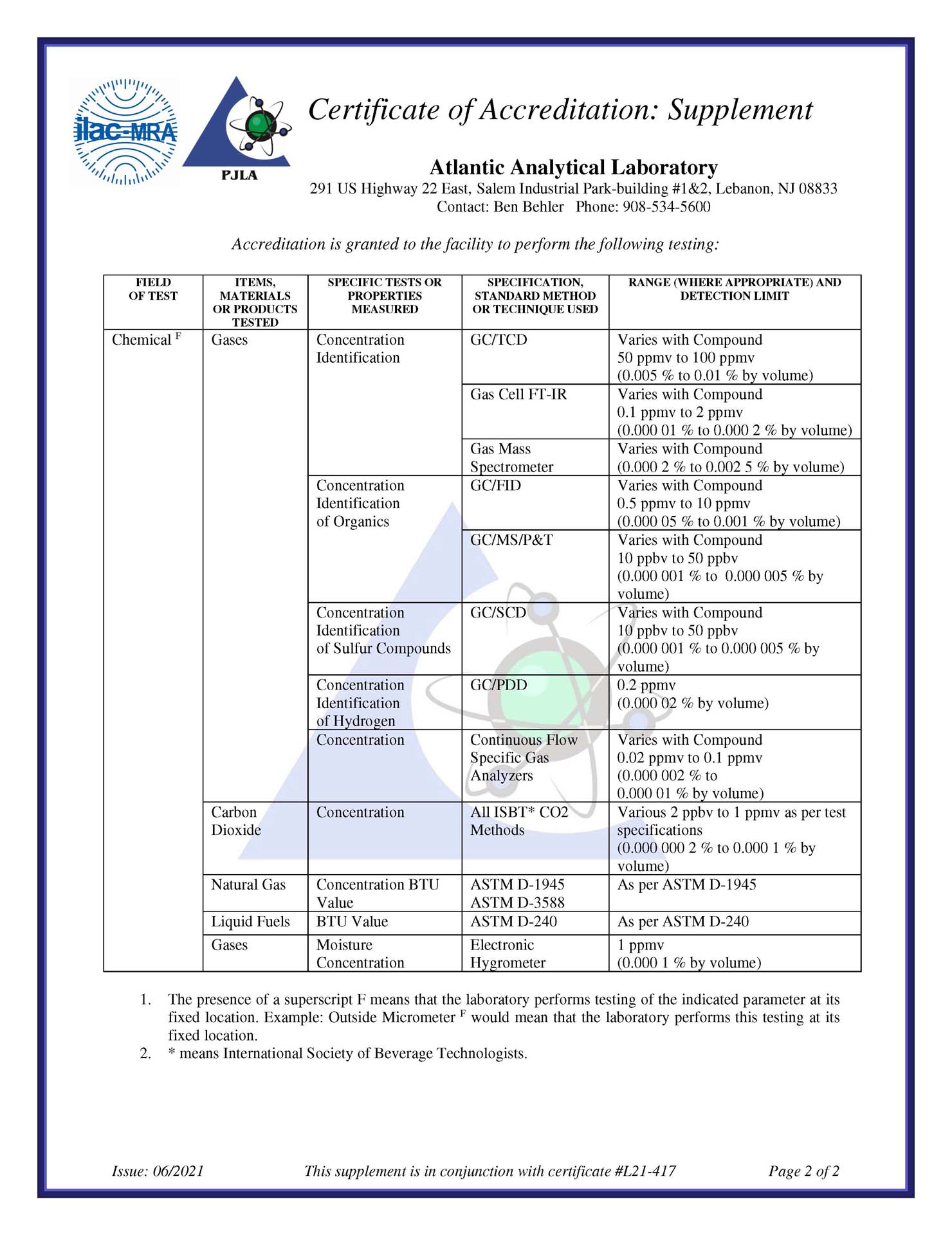
We routinely follow the current monographs for USP/NF and EP testing for Nitrogen, Oxygen, Carbon Dioxide, Air, and Argon, providing Certificates of Analysis for:
Visit our contact page or give us a call today to get started!
- NF Nitrogen
- NF Nitrogen – 97%
- EP Nitrogen
- EP Nitrogen, Low Oxygen
- EP Oxygen
- EP Argon
- USP Oxygen
- USP Oxygen – 93%
- USP Carbon Dioxide
- USP Medical Air
- USP Helium
USP, NF & EP Gas Testing FAQ
USP, NF, and EP gas testing refers to analytical testing performed according to the current monographs published by:
United States Pharmacopeia (USP)
National Formulary (NF)
European Pharmacopoeia (EP)
These compendia establish official quality standards for pharmaceutical substances, including medical and process gases.
While all three compendia establish pharmaceutical quality standards, they apply to different regulatory regions and product classifications:
USP: Commonly used in the United States for drug substances and medical gases
NF: Focuses primarily on excipients and certain pharmaceutical ingredients
EP: Applies to products marketed in the European Union and other regions recognizing the European Pharmacopoeia
Each monograph may specify different purity thresholds, impurity limits, and analytical methods.
Testing frequency depends on regulatory requirements, internal quality systems, and product release procedures. Many clients perform testing:
Per batch
Per lot
At defined release intervals
We can help determine an appropriate testing schedule based on your quality program.